ISIS Neutron and Muon Source Data Journal
This is a page describing data taken during an experiment at the ISIS Neutron and Muon Source. Information about the ISIS Neutron and Muon Source can be found at https://www.isis.stfc.ac.uk.
Entropy change of protein and water during the denaturation of cytochrome P450s
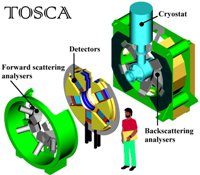
Abstract: Cytochrome P450s are ubiquitous hemeprotein monooxygenases that catalyse numerous biochemical reactions, and the investigation on them is of significance to both the protein engineering and human health. We found that the thermophilic cytochrome P450, CYP119, may achieve both greater thermal stability and higher structural flexibility simultaneously by keeping larger entropy at the native state. Although we have measured the thermodynamic curves during the denaturation of both CYP101A1 and CYP119, it�s quite difficult to distinguish the entropy change of protein and water. We believe water molecules play an important role of protein stability, especially in the functional state. Fortunately, combining inelastic neutron scattering and protein perdeuteration, we may obtain the density of states of protein and water, respectively, and calculate the entropy change.
Public release date: 01 June 2024
Principal Investigator: Professor liang hong
Experimenter: Mr Zhuo Liu
Experimenter: Mr Lirong Zheng
Experimenter: Mr Lei Zhang
Local Contact: Dr Svemir Rudic
DOI: 10.5286/ISIS.E.RB1920133-3
Parent DOI: 10.5286/ISIS.E.RB1920133
ISIS Experiment Number: RB1920133
Part Number: 3
Date of Experiment: 27 May 2021
Publisher: STFC ISIS Neutron and Muon Source
Data format: RAW/Nexus
Select the data format above to find
out more about it.
Data Citation
The recommended format for citing this dataset in a research
publication is as:
[author], [date], [title], [publisher],
[doi]
For Example:
Professor liang hong et al; (2021): Entropy change of protein and water during the denaturation of cytochrome P450s, STFC ISIS Neutron and Muon Source, https://doi.org/10.5286/ISIS.E.RB1920133-3
Data is released under the CC-BY-4.0 license.